Adverse Drug Reactions | Vibepedia
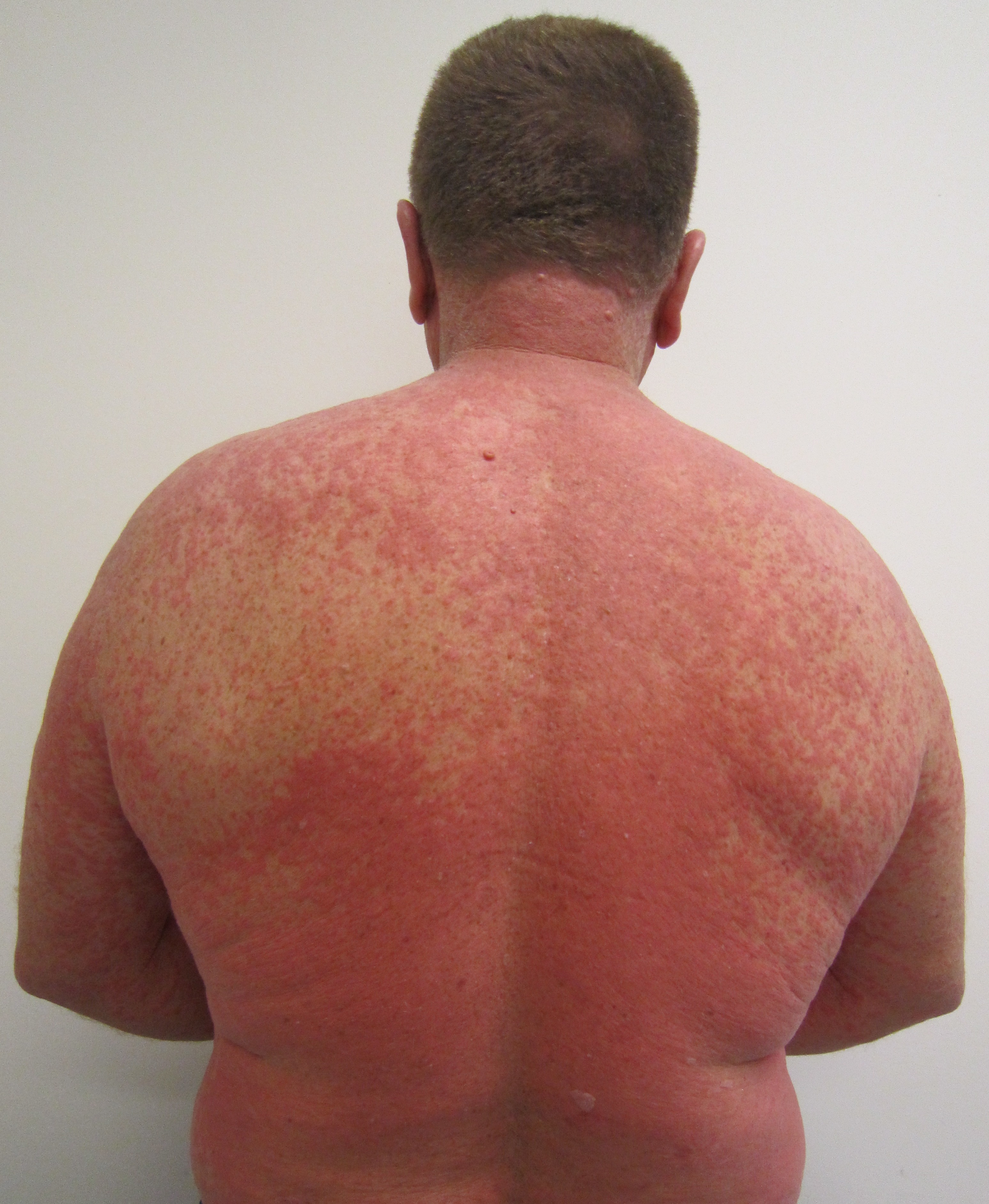
Adverse Drug Reactions (ADRs) are unintended and often harmful responses to a medication. These reactions range from mild discomforts like nausea to…
Contents
Overview
The recognition of unintended harm from medicines predates formal scientific study, with ancient texts hinting at the dual nature of potent substances. The systematic study of Adverse Drug Reactions (ADRs) gained significant momentum in the mid-20th century, spurred by catastrophic events like the thalidomide disaster. The Kefauver-Harris Amendment mandated proof of efficacy and safety before marketing. This era also saw the establishment of early pharmacovigilance systems, laying the groundwork for the comprehensive monitoring we see today, driven by institutions like the World Health Organization's Uppsala Monitoring Centre.
⚙️ How It Works
ADRs arise from a complex interplay between a drug's pharmacological properties and an individual's unique biological makeup. ADRs can be broadly categorized into Type A (augmented), which are dose-dependent and predictable based on the drug's known mechanism of action (e.g., bleeding from anticoagulants like warfarin), and Type B (bizarre), which are unpredictable, not dose-dependent, and often immune-mediated or idiosyncratic (e.g., severe allergic reactions to penicillin). Other classifications of ADRs include Type C (chronic), Type D (delayed), and Type E (end of use). The underlying mechanisms can involve direct toxicity, hypersensitivity reactions, genetic predispositions (pharmacogenomics), drug-drug interactions, or even interactions with food or the environment, all contributing to the diverse spectrum of patient responses to pharmaceuticals.
📊 Key Facts & Numbers
Globally, ADRs are a major public health concern. These figures highlight the pervasive impact of unintended drug effects across diverse healthcare systems and patient populations.
👥 Key People & Organizations
Pioneering figures in pharmacovigilance and drug safety have been instrumental in understanding and mitigating ADRs. The Bradford Hill criteria are a set of guidelines used to assess the likelihood that a suspected adverse event is caused by a medication. Regulatory bodies like the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA) are central to monitoring ADRs. Pharmaceutical companies themselves employ dedicated pharmacovigilance departments to track and report these events for their products, such as those developed by Pfizer and Merck. Organizations like the World Health Organization (WHO) also play a crucial role in global coordination.
🌍 Cultural Impact & Influence
The cultural impact of ADRs is profound, shaping public trust in medicine and influencing healthcare policy. The thalidomide crisis fundamentally altered perceptions of pharmaceutical safety and led to stricter regulatory oversight, impacting how drugs are developed and marketed. Public awareness campaigns and patient advocacy groups, such as those focused on autoimmune conditions or oncology treatments, often highlight the risks and realities of ADRs. Media portrayals of medical dramas and news reports frequently feature stories of unexpected drug reactions, contributing to both informed caution and, at times, undue fear surrounding medication use. The ongoing dialogue about ADRs influences patient-doctor communication and shared decision-making in medical treatment.
⚡ Current State & Latest Developments
Current efforts in ADR management focus on leveraging advanced technologies and data analytics. Artificial intelligence (AI) and machine learning are increasingly employed to sift through vast datasets from electronic health records, social media, and spontaneous reporting systems to identify potential ADR signals earlier and more efficiently than traditional methods. Precision medicine, guided by pharmacogenomic testing, aims to predict individual susceptibility to ADRs based on genetic profiles, allowing for personalized drug selection and dosing. Furthermore, initiatives like the U.S. Food and Drug Administration's Sentinel Initiative utilize real-world data to monitor drug safety post-approval, moving beyond passive reporting to active surveillance.
🤔 Controversies & Debates
Significant controversies surround the reporting and management of ADRs. Spontaneous reporting systems for ADRs are known to be prone to underreporting, bias, and a lack of detailed clinical information. Critics argue that regulatory agencies and pharmaceutical companies may downplay or delay acknowledging serious ADRs, particularly for blockbuster drugs, due to economic or reputational concerns. The definition and classification of ADRs themselves can also be contentious, with ongoing discussions about how to best distinguish between true ADRs, side effects, and other patient outcomes. The balance between drug benefit and risk is a constant ethical and scientific challenge, particularly for novel therapies like gene therapies or mRNA vaccines.
🔮 Future Outlook & Predictions
The future of ADR management points towards increasingly proactive and predictive strategies. The expansion of pharmacogenomic databases and their integration into clinical practice will enable more personalized and safer prescribing. Furthermore, the development of 'digital twins' – virtual replicas of patients – could allow for simulated drug trials to predict individual responses and ADRs. Regulatory agencies are also exploring real-world evidence (RWE) more extensively for post-market surveillance, potentially leading to faster interventions when safety signals emerge.
💡 Practical Applications
The practical applications of understanding ADRs are vast and critical for patient care. Healthcare professionals use ADR information to select appropriate medications, adjust dosages, and monitor patients for potential adverse effects. Pharmacists play a key role in counseling patients about potential ADRs and identifying drug interactions. Public health initiatives leverage ADR data to inform prescribing guidelines and drug safety warnings. For patients, understanding ADRs empowers them to communicate effectively with their healthcare providers about their experiences and potential risks associated with their medications, contributing to safer and more effective medical outcomes.
Key Facts
- Category
- science
- Type
- topic