Metal on Metal Hip Implant Failures | Vibepedia
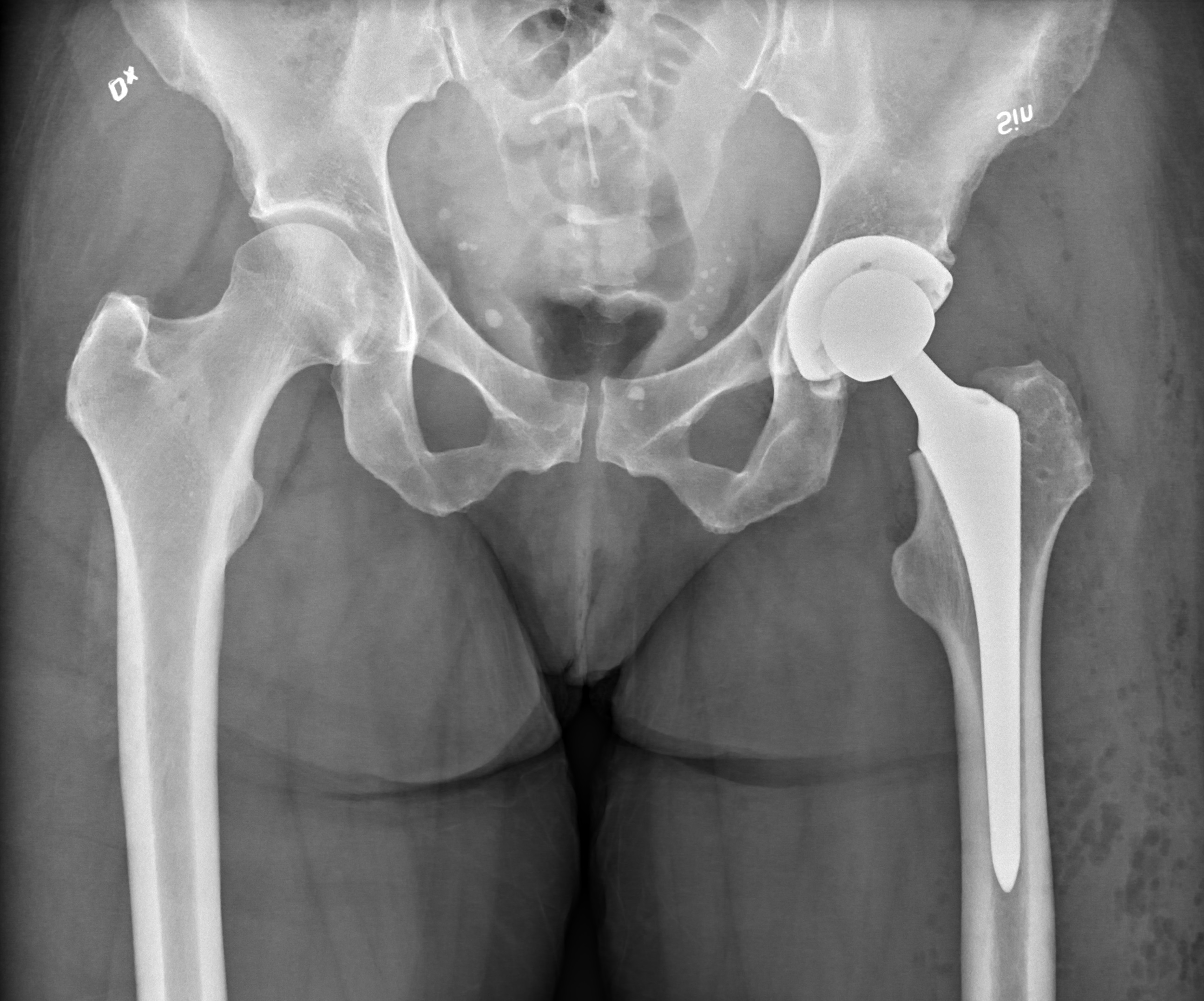
Metal-on-metal (MoM) hip implants, once hailed as durable solutions for hip joint degeneration, became a significant public health crisis due to widespread…
Contents
Overview
Metal-on-metal (MoM) hip implants, once hailed as durable solutions for hip joint degeneration, became a significant public health crisis due to widespread failures. These devices featured articulating surfaces made entirely of metal, designed to mimic natural hip movement. However, the constant friction between the metal components released microscopic metal particles and ions into the surrounding tissues and bloodstream. This metallic debris led to a cascade of adverse reactions, including metallosis (tissue damage from metal accumulation), pseudotumors (fluid-filled masses), and severe systemic toxicity. Thousands of patients experienced debilitating pain, mobility loss, and required painful revision surgeries. The failures prompted numerous lawsuits, regulatory scrutiny, and a dramatic shift away from MoM technology in hip arthroplasty, leaving a legacy of patient suffering and a cautionary tale for medical device innovation.
🎵 Origins & History
The Birmingham Hip Prosthesis was an early metal-on-metal hip implant design. These implants were envisioned as a superior alternative to earlier materials like polyethylene and acrylic bone cement, promising greater longevity and reduced wear. Companies like DePuy Orthopaedics (a subsidiary of Johnson & Johnson) and Biomet heavily invested in developing and marketing MoM systems throughout the 1990s and early 2000s. The allure was the perceived durability and the potential for larger femoral heads, which could offer greater stability and range of motion. This era saw a resurgence of interest in MoM designs, with many manufacturers reintroducing or refining their metal-bearing surfaces, often without sufficient long-term clinical data to support their safety and efficacy beyond a decade.
⚙️ How It Works
Metal-on-metal hip implants function by having two primary articulating surfaces made of metal: the femoral head (the ball) and the acetabular cup (the socket). As the patient moves their hip, these metal surfaces grind against each other. Ideally, this friction would generate minimal wear debris. However, in practice, the constant articulation releases microscopic metal particles, primarily cobalt and chromium, into the joint space. These particles can cause a severe inflammatory response in the surrounding tissues, leading to a condition known as metallosis. Metallosis is tissue damage from metal accumulation. The accumulation of metal ions can also enter the bloodstream, potentially causing systemic effects. In many cases, this inflammatory reaction leads to the formation of pseudotumors, which are abnormal masses of tissue that can compress nerves and blood vessels, causing significant pain and functional impairment. Pseudotumors are fluid-filled masses that can form due to metal debris.
📊 Key Facts & Numbers
Estimates suggest that the use of metal-on-metal hip implants has drastically declined. The U.S. Food and Drug Administration (FDA) has implemented stricter guidelines for the approval of new hip implant designs. Ceramic-on-ceramic and highly cross-linked polyethylene are alternative bearing surfaces for hip implants. 3D printing technologies are being leveraged in the development of next-generation implants.
👥 Key People & Organizations
Key figures in the MoM hip implant saga include Dr. William McAlpin, an orthopedic surgeon who championed early MoM designs, and Dr. Charles Bullock, who later became a vocal critic, highlighting the adverse tissue reactions. Companies like DePuy Orthopaedics (with its A.S.R. Hip System) and Biomet (with its M2a Magnum) were central manufacturers whose products faced significant litigation. Regulatory bodies such as the U.S. Food and Drug Administration (FDA) and the Medicines and Healthcare products Regulatory Agency (MHRA) in the UK played crucial roles in investigating and responding to the failures. Patient advocacy groups, though often fragmented, also emerged to share experiences and push for accountability.
🌍 Cultural Impact & Influence
The MoM hip implant crisis contributed to a broader public discourse on patient safety and informed consent. The thousands of lawsuits filed against companies like DePuy Orthopaedics and Biomet generated significant media attention, often featuring harrowing patient testimonials. This crisis contributed to a broader public discourse on patient safety, informed consent, and the ethical responsibilities of the medical industry. The narrative of MoM implants shifted from a technological marvel to a cautionary tale, influencing how patients and physicians approach surgical options and the perceived risks associated with new medical technologies. The legacy of MoM failures continues to shape patient expectations and physician decision-making in orthopedic surgery.
⚡ Current State & Latest Developments
Following the wave of recalls and litigation, the use of metal-on-metal hip implants has drastically declined. Most major manufacturers have ceased production or significantly reformulated their MoM offerings. Regulatory bodies like the U.S. Food and Drug Administration (FDA) have implemented stricter guidelines for the approval of new hip implant designs, including requirements for longer-term clinical data and enhanced post-market surveillance. While some MoM implants remain in use, the focus has shifted to alternative bearing surfaces such as ceramic-on-ceramic and highly cross-linked polyethylene. The orthopedic industry is now heavily invested in developing next-generation implants with improved biocompatibility and wear resistance, often leveraging advanced materials and 3D printing technologies.
🤔 Controversies & Debates
The primary controversy surrounding MoM hip implants centers on whether manufacturers adequately tested these devices before widespread release and whether they responded appropriately to early warning signs of failure. Critics argue that companies prioritized profit over patient safety, downplaying adverse event reports and continuing to market potentially dangerous products. The debate also extends to regulatory bodies, with questions raised about the effectiveness of pre-market approval processes and post-market surveillance. Furthermore, there's ongoing discussion about the long-term health implications of chronic metal ion exposure, with some studies suggesting potential links to neurological issues and certain cancers, though definitive causal relationships remain debated and require further research.
🔮 Future Outlook & Predictions
The future of hip implant technology is likely to move further away from metal-on-metal designs. Expect continued innovation in ceramic-on-ceramic and highly cross-linked polyethylene bearing surfaces, aiming for even lower wear rates and enhanced biocompatibility. The integration of robot-assisted surgery and artificial intelligence in implant placement is expected to improve precision and outcomes. Personalized implants, potentially designed using patient-specific imaging data and 3D printing, could become more common, offering tailored solutions for complex anatomical variations. The focus will remain on minimizing wear debris, reducing inflammatory responses, and achieving long-term implant survival with minimal patient complications, learning from the costly lessons of the MoM era.
💡 Practical Applications
The primary practical application of understanding metal-on-metal hip implant failures is in patient education and informed consent. For individuals considering hip replacement surgery, awareness of the historical issues with MoM implants is crucial for making informed decisions about implant bearing surfaces. It also informs orthopedic surgeons about the risks and benefits of different implant types, guiding them toward safer, more established technologies. Furthermore, the failures have spurred the development of better diagnostic tools for monitoring implant health, including blood tests for metal ion levels and advanced imaging techniques to detect metallosis and pseudotumors. This knowledge directly impacts the clinical management of patients with existing MoM implants, facilitating timely intervention when complications arise.
Key Facts
- Category
- technology
- Type
- topic